Introduction
Gemmotherapy, also known as phytoembryotherapy, is an alternative phytotherapeutic treatment that uses extracts prepared mainly from the embryonic tissues of plants, e.g., emerging shoots and buds of various trees and shrubs. This type of therapy is used in European countries, such as Germany, France, Belgium, and Italy [1] [2]. The herbal drug is typically collected during spring, at the peak time of the tree’s or shrub’s germination. Gemmotherapy claims, that shoots and buds are exceptional parts of a plant in space and time. They develop substances not contained in the adult plants and provide high concentrations of minerals, vitamins, plant hormones, etc. [2]. It also claims to have higher biological activity in comparison to other traditional treatments with herbal drugs [2]. For preparation of the gemmotherapy medicines, the fresh buds or shots of the plant are macerated for 21 days with a mixture of alcohol and glycerine, at a ratio of 1:20 fresh plant material : mixture. The extracts are then filtered and diluted 1:10 with a mixture of glycerine, alcohol and water [3].
In her high school diploma work, Ms. Brunner qualitatively investigated the chemical differences between the fingerprints of the buds and mature leaves of four herbal drugs used in gemmotherapy: birch (Betula pubescens EhRh.), hazel (Corylus avellana L.), ash (Fraxinus excelsior L.), and dog rose (Rosa canina L.). Samples of each specimen have been collected at different germination periods between spring (when sprouting started) and summer of 2019 (mature leaves). The focus of the investigation was on secondary metabolites, which are generally considered as active principle of herbal drugs. Ms. Brunner was honoured with the “Simply Science” prize of the “Schweizer Jugend forscht” contest in 2020 [4].
Standard solutions
System Suitability Test (SST): verbascoside and quercitrin are prepared in methanol at concentrations of 1 mg/mL and 0.5 mg/mL, respectively.
Sample preparation
After collection, the samples are kept in the freezer to preserve freshness. Prior to extraction, any ice crystals on the frozen buds or leaves are removed mechanically. Then, the material is cut into small pieces with a knife. For the extraction, 500 mg of the cut material are mixed with 5mL of methanol and sonicated for 10minutes at room temperature. The mixture is centrifuged for 5 minutes and the supernatant is used as test solution.
Chromatogram layer
HPTLC plates silica gel 60 F254 (Merck), 20 x10 cm
Sample application
Automatic TLC Sampler (ATS 4), 15 tracks, band length 8.0 mm, track distance 11.4 mm, distance from left edge 20.0 mm, distance from lower edge 8.0 mm, application volume 2.0 μL
Chromatography
In the ADC 2 with chamber saturation for 20 min (with filter paper) and after conditioning at 33% relative humidity for 10 min using a saturated solution of magnesium chloride, development with ethyl acetate, formic acid, water (15:1:1 v/v/v) to 70 mm (from the lower edge), drying for 5 min
Post-chromatographic derivatization
The plate is heated at 100 °C for 3 min using the TLC Plate Heater, immersed into natural product (NP) reagent (1.0 g of 2-aminoethyl diphenylborinate dissolved in 200 mL of ethyl acetate) dried under cold air and documented. Subsequently, the plate is immersed into anisaldehyde-sulfuric acid (AS) reagent (1.0mL of p-anisaldehyde dissolved in 200 mL of a mixture of methanol, acetic acid and sulfuric acid 17:2:1 (v/v/v)) and then heated at 100 °C for 3 min.
Documentation
TLC Visualizer 2 under UV 254 and UV 366 nm (prior to derivatization), UV 366 nm (after derivatization with NP reagent) and white light (after derivatization with NP plus AS reagents)
Results and discussion
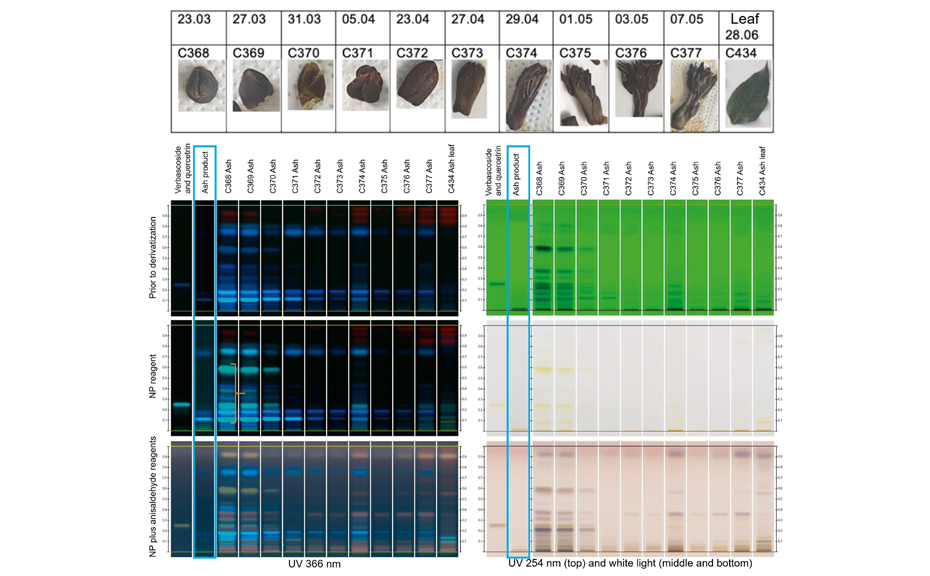
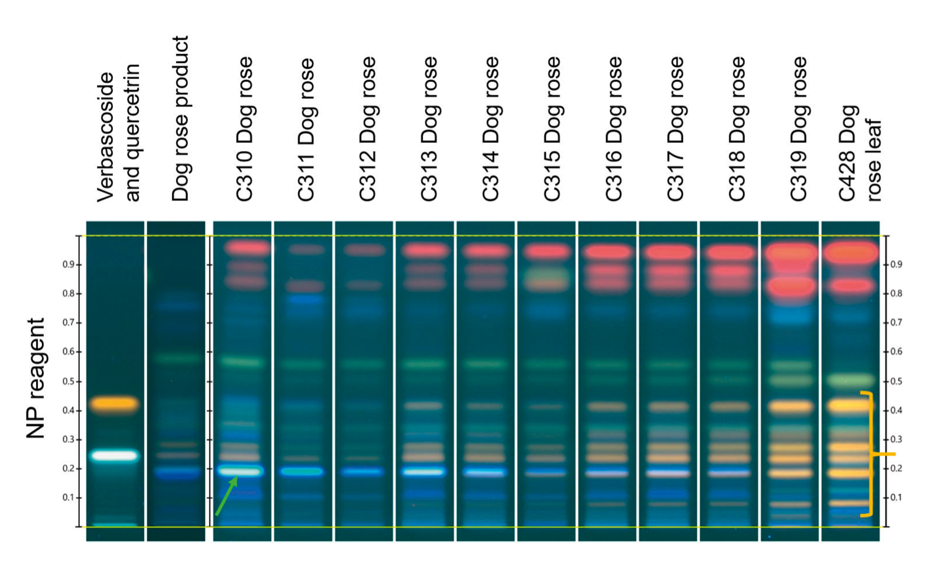
HPTLC analyses of birch, ash, hazel and dog rose samples were performed with samples from three individual plants of each species, collected in the Basel area in Switzerland. The buds and leaves of these plants were gathered eleven times between March and June of 2019. Results are shown in the figures below and are reported for one individuum of ash and dog rose. Their fingerprints were compared to those of the respective gemmotherapy products.
The following observations were made: first, the fingerprints of the buds are very different from those of the mature leaves, e.g., the young bud of ash (C368) and its respective mature leaf (C434), under several detections (see shown figures). Second, the younger buds contain generally a higher concentration of secondary metabolites than buds with first shots (leaves) or, in some cases, the matured leaves. For example, samples of young bud of ash (C368, C369, C370) show several blue intense zones under UV 366 nm after derivatization with NP (orange bracket), corresponding to hydroxycinnamic acid derivatives, not seen in the subsequent samples. Third, the transition from buds to young leaves is generally paralleled by a gradual change in the composition of the fingerprints. For example, the more mature the buds, the lower the content of hydroxy-cinnamic acid derivatives. In the case of dog rose, a decrease is observed in the content of hydroxy-cinnamic acid derivatives (green arrow) accompanied by an increase in the number of zones corresponding to flavonoids (yellow zones, yellow bracket).
The fingerprints of commercial gemmotherapy products show similarities with those of the buds, but fingerprints are generally fainter (e.g. blue frame). This was expected, because the products are usually prepared in a diluted form, following the homeopathic philosophy.
Additional experiments will be performed to investigate the variation in the composition more quantitatively and to remove the freezing process prior to extraction, in order to copy the industrial preparations for gemmotherapy.
[1] A.-D. Raiciu, “Gemmotherapy – Modern Medicine,” in Proceedings, 2019, vol. 29, p. 117.
[2] A. Sarkova and M. Sarek, “EAV and Gemmotherapy – Medicine for the Next Millennium? (technique as a means to link eastern and western medicine),” in Engineering in Medicine and Biology 27th Annual Conference, 2005, pp. 4943–4946.
[3] Seroyal, “Gemmotherapy. Embryonic tissue extracts to stimulate organ systems toxin elimination toward the emunctories,” Reference guide. [Online]. Available: https://www.seroyal.com/media/wysiwyg/seroyal/downloads/Gemmotherapy_Brochure.pdf. (accessed: 17.07.2020).
[4] https://sjf.ch/arbeiten-nationaler-wettbewerb/ (accessed: 20.07.2020)
Further information is available on request from the authors.
Contact: Débora Frommenwiler, CAMAG, Sonnenmattstrasse 11, 4132 Muttenz, Switzerland, debora.frommenwiler@camag.com