Our Vision & Strategy
To create value for our customers by providing instruments, analytical and diagnostic methods and training to analysts worldwide, to allow the direct analysis of diverse samples gained directly from complex matrices by means of instrumental High-Performance Thin-Layer Chromatography (HPTLC) or of instrumental Dried Matrix Spot Extraction (DMS); successfully implemented in case the use of these methods produce the best possible analytical or diagnostic result, at a competitive price and leaves a satisfied customer. CAMAG creates, tests and produces all instruments solely in Switzerland - “SWISS MADE”, and provides them to its worldwide customers through its subsidiaries and dedicated distributors (more than 70).
Our History
CAMAG was founded by the German Chemist Dr. Dieter Jänchen (1927-2018) in 1958 as “Chemie-Erzeugnisse und Adsorptionstechnik Muttenz AG”. The first product produced was activated Aluminium oxide, still sold today in bulk quantities. In 1961, CAMAG entered the analytical field of Thin-Layer Chromatography (TLC). We extended our market position over the years to become the world leader in instrumental High-Performance Thin-Layer Chromatography (HPTLC). In most analytical laboratories in the world, you will find at least one of our instruments. Lately, CAMAG has entered the diagnostics field by developing a fully automated, high-throughput device for the metabolite extraction of Dried Matrix Spot cards.
CAMAG has started to launch its fully automated next generation HPTLC instruments in September 2019 under the name of CAMAG® HPTLC PRO SYSTEM. Module APPLICATION and Module DEVELOPMENT have been launched, the other Modules will be launched sequentially over the next two years. Then, the first fully automated sample analysis and evaluation system for routine quality control in HPTLC analysis will exist. New patents speak for the value of this innovation, leaving customers with even improved cost efficiency.

CAMAG® HPTLC PRO
Innovation will always lead the path of CAMAG.
The decade of CAMAG®’s HPTLC Software visionCATS. The first version was launched in 2012. This software established HPTLC over TLC. It stands for ease of use and intuitive simplicity. The software organizes the workflow of HPTLC and controls all involved CAMAG® instruments. The easy-to-navigate user interface guides effectively through the chromatographic process - from analysis definition to analysis reporting. As state-of-the-art software, visionCATS is based on a client-server system offering enormous flexibility to the number of instruments and users that are working together, and enabling access to the same data for all members of a work group. The sample-oriented approach allowed for creating virtual plates with tracks originating from different plates, for example, batch-to-batch comparison or long-term stability testing. Further new functionality was added with visionCATS: a powerful search tool and a file explorer, which includes extended preview functionalities, enable highly comfortable searches for samples, methods, and analysis files.

CAMAG® HPTLC Software visionCATS
In 2009, CAMAG launched the CAMAG® TLC-MS Interface based on the Luftmann patent (DE10036293C2). This device is a versatile instrument to elute compounds from a TLC/HPTLC plate and feed them into a Mass Spectrometer for substance identification or structure elucidation. In 2016, the optimized successor – CAMAG® TLC-MS Interface 2 – was released.

CAMAG® TLC-MS Interface 2
In 2012, CAMAG entered the diagnostic field by developing a patented, fully automated, high-throughput device – the CAMAG® DBS-MS 500 – for the metabolite extraction of Dried Matrix Spot cards. The knowhow gained from the elution process of the TLC-MS Interface was transferred to this device to extract target analytes online from Dried Matrix Spots and analyze directly with LC-MS or MS, or to collect by a Sample Collector.

CAMAG® DBS-MS 500
Another patented break-through took place in 2016 with the first automated spraying device for reagent transfer on TLC/HPTLC plates – the CAMAG® Derivatizer. This instrument was the missing piece to be able to automate all HPTLC steps. With its unique and patented micro-droplet technology, a fine reagent aerosol is generated by a nozzle. The reagent is evenly distributed in the chamber and settles gradually onto the plate.
In 2016, two successors of instruments were released, the CAMAG® UV Lamp 4 (together with the CAMAG® UV Cabinet 4) and the CAMAG® TLC Visualizer 2.
The next level of the software was released in 2000 with a new concept: winCATS Planar Chromatography Manager, whose development was already started in the mid-1990s. As the name says, all main CAMAG devices were controlled with this software and all data and results were saved according to cGMP. winCATS offered the option for 21 CFR Part 11 compliance and was worldwide unique in terms of concept, scope and quality in our analytical field. The customer could define the extent of documentation. For all individual devices, SOPs (Standard Operation Procedures) were developed and specially trained CAMAG specialists could now carry out IQ/OQ (Installation Qualification/Operation Qualification) certifications on customer's request.
At the same time, CAMAG launched the CAMAG® Automatic TLC Sampler 4 (ATS 4), which is still state-of-the-art today. Precision and robustness of the sample application in the nL and μL range were further increased. In addition to spot and band application, the possibility of application of matrix and water-containing samples in the form of a rectangle was added.
In 2001, the Linomat family was advanced with the CAMAG® Linomat 5 (still state-of-the-art), which is operated with our software.
From 2005 on, industrial digital cameras revolutionized CAMAG´s documentation system. Initially, DigiStore 2 combined the Reprostar 3 lighting unit with a high-resolution, high-linear 12-bit CCD camera and data transfer via FireWire. The system combined functions for exposure optimization and background correction with excellent color fidelity.
In 2002, the bioluminescence detection for activity and toxicity screening – initially based on a test developed by Bayer using the marine bacteria Vibrio fischeri (reclassified later to Allivibrio fischeri) – was added to the product range, and was optimized in 2006 with the release of the CAMAG BioLuminizer®. A cooled 16-bit CCD camera with high resolution and quantum efficiency still allows the inexpensive and fast detection of sample components with biological activity.
Since 2005, the CAMAG® ADC 2 offers automated development of HPTLC plates (20 x 10 cm). The automated process ensures the preconditioning and activity setting of the layer, the monitoring of the development process by means of a CCD sensor, and the control of the drying conditions. With this in Planar Chromatography a previously unattained consistency of the RF values – within a plate and from plate to plate – is achieved. The ADC 2 was initially operated via winCATS, now with visionCATS or stand-alone.
With the use of high-quality industrial digital cameras for documentation and image-based evaluation, the demands on the illumination unit increased. CAMAG® TLC Visualizer is since 2008 the corresponding improved solution.

CAMAG® TLC Visualizer
Our CATS software was enhanced with a considerable number of user-friendly features and options, including various calibration functions, path optimization, background subtraction, spectrum acquisition, spectrum library, to name but a few. CATS also supported the calculation of results in mg/tablet, Content Uniformity Test (CUT), and the correlation calculation of the corresponding spectra for identity and purity tests, which is important for many pharmaceutical customers.
CAMAG took into account the requirements of cGMP (current Good Manufacturing Practice) with its equipment and software development.
Starting in 1991, CAMAG established a quality assurance system, on the basis of which the company achieved ISO 9001 certification at its first attempt in 1995 and has since regularly regained it.
With the CAMAG® Automatic Sample Applicator III (ATS III), a software-controlled device was introduced to the market in 1990 that enabled spot and band-shaped application in the nL and μL range. Initially, it was operated stand-alone. Later, the ATS 3 software was integrated into CATS so that the corresponding parameters did not have to be entered again for the Scanner. The ATS III helped many customers worldwide to use TLC and HPTLC for quantitation. The device remained in the CAMAG product range for 10 years until it was replaced by the ATS 4 in 2000.

CAMAG® ATS III
The slightly less demanding sample applicator, CAMAG® Linomat IV, was introduced at the beginning of this decade and remained in the program until 2001. The device was microprocessor controlled, the application parameters were entered via a keyboard.
In 1992, with the CAMAG® Automatic Developing Chamber (ADC), the development step was automated and thus made convenient for the user.
In 1994 we launched the CAMAG® TLC Scanner 3. The device features a spectral range from 190 to 800 nm, a measuring speed of up to 100 mm/s, step resolution selectable between 25 and 200 μm, and fast spectra acquisition of up to 100 nm/s at an oversampling rate of 40/nm.
Despite all the advances in classical densitometry and the advantages of spectral resolution, it became apparent that many users of Planar Chromatography wanted to use electronic image acquisition either alternatively or complementarily. Therefore CAMAG extended its documentation system with Video-Store for documentation, archiving and with VideoScan for quantitation.
We should also mention CAMAG® AMD 2, which we released in 1997. With this system, the development paths are no longer controlled by time increments, but, as with the ADC, directly by a CCD sensor. AMD is still the only way for many users to separate matrix-loaded multicomponent mixtures with high resolution using HPTLC.
When we moved into our new building in the Sonnenmattstrasse in Muttenz in 1980, the CAMAG® TLC Scanner (I) had become our most successful device in the product range. We had initially built a series of 10 and planned to produce a few more small series before it would be replaced by a successor model. In fact, we sold more than 500 units before we released our CAMAG® TLC-Scanner II in 1983.
The Scanner was mainly operated in connection with an integrator, partly only with an analog recorder. As early as 1976, Siegfried Ebel and his group in Marburg, later Würzburg, equipped the Zeiss KM3 Scanner with a computer for operation. As soon as the CAMAG Scanner was on the market, Ebel developed a computer control system for it as well. From 1980 CAMAG sold Ebel's software, later we developed our own.
Sample application was now the step that had to be automated. European competitors in the TLC market had various solutions for this. All of them were based on the fact that parallel dosing devices applied several samples simultaneously. Desaga used a peristaltic pump, Shandon and CAMAG syringes operated in parallel. The CAMAG device had the special feature that the expansion of the application zone was controllable.

CAMAG® Automatic Sample Applicator for conventional TLC plates (1974)
It turned out that the principle of parallel application was not suitable for the miniaturized dimensions of HPTLC, so we developed the CAMAG® TLC Sampler I. It used a single dosage module with which the samples fed from a rack were sequentially applied to a plate. The device was software-controlled and was suitable for both conventional TLC and HPTLC plates.
For the CAMAG® TLC Scanner II we first developed our software 86 and later CATS (CAMAG® TLC Software). With the concept of this software and various evaluation options, a high degree of acceptance on the market was achieved over many years.

CAMAG® TLC Scanner II (1983)
Klaus Burger, Bayer Dormagen, had completed the Programmed Multiple Development process presented by Perry, Glunz et al. in the 1970s to a multi-stage gradient development. We constructed a marketable system, which we presented to the public at the International Symposium on HPTLC in Würzburg (1983) under the name of AMD (Automated Multiple Development). The principle introduced a completely new, previously unknown separation performance and application areas to HPTLC.
The list of these device developments should not make us forget that we also brought out a whole series of less spectacular innovations, including CAMAG® Linomat IV, the CAMAG® Reprostar II photo documentation system and various further developments of our product range.
This period includes the systematic instrumentation of all TLC steps. This became inevitable when Merck launched the HPTLC plates in 1975. CAMAG became a pioneer in instrument development and subsequently the recognized market leader in Thin-Layer Chromatography.
At the beginning of this decade, experts did not yet agree whether quantification by direct photometric evaluation or by elution of the separated zones with subsequent photometry in solution leads to more precise results. CAMAG® Eluchrome allowed for the elution of six substance zones with 1.5 mL solvent in parallel without scraping the layer off the plate.

CAMAG® Eluchrome
With the CAMAG® Linomat, the corner stone was laid for a family of application devices that use spray-on technique for sample application. The previously used capillaries for spot application often destroyed the layer surface. Also, the spray-on application of bands significantly improved the separation performance of TLC. While the first two generations of Linomat were still predominantly used for preparative tasks, the CAMAG® Linomat III (1976) significantly improved exact dosing and reproducibility, as required for quantitative TLC/HPTLC.
Since 1973, CAMAG provides TLC training courses, initiated by Rudolf E. Kaiser and initially held at his Institute for Chromatography in Bad Dürkheim, Germany. The German company Merck presented new plates at one of those courses in 1975, and Kaiser immediately devised a revolutionary circular development technique for these plates. Within two months, CAMAG developed the saleable U-chamber from Kaiser’s functional prototype. We were able to pull this off using the high-precision fluid metering device developed for our Linomat III, whose own development was close to completion.

CAMAG® Linomat III

CAMAG® U chamber
The new HPTLC layers required additional care during sample application, regardless of whether circular or linear chromatography was applied. CAMAG developed the CAMAG® Nanomat (I). PtIr capillaries were lowered magnetically onto the layer to apply fixed sample volumes of 100 or 200 nL. As an alternative option to dose variable volumes of 50-230 nL, we offered the Nanoapplicator (combination of a syringe with a micrometer screw) which could be integrated into the Nanomat.
In 1976, a new chamber with horizontal plate position especially for HPTLC layers was released. This "linear development chamber" later became the Horizontal Development Chamber, which is still available today. This chamber allows for chromatogram development from both plate edges to the middle, doubling the number of samples, and requires only a few mL of solvent.
The culmination of this decade was the invention of the first CAMAG® TLC Scanner, which we launched in 1978. The spectral range was 200-800 nm. It was designed for absorption and fluorescence measurements in remission and transmission, the latter because we wanted to make it universally usable, both for the evaluation of Thin-Layer Chromatograms and electropherograms. The Scanner was also set up for the evaluation of circularly developed chromatograms. It was available in two versions, one equipped with a filter wheel and one with a more expensive monochromator. On one hand, we built the latter in order not to be inferior to the competing instrument Zeiss KM 3, on the other hand we thought the market would opt for the cheaper filter version – a glaring misjudgement! The CAMAG® TLC Scanner I contributed significantly to the breakthrough of quantitative TLC.

CAMAG® TLC Scanner I
The original purpose of the company was the production of Aluminium Oxide for chromatography. It was offered with different pH values (basic, neutral, acidic), and mainly sold at the highest activity levels according to Brockmann, i.e. Brockmann activity I. Applications ranged from Preparative Column Chromatography on a laboratory scale to the industrial use, mainly for the removal of by-products from the compound of interest. Today, CAMAG is still a reliable producer of Aluminium Oxide and has a REACH accreditation for Europe.
The foundation of CAMAG coincided with the rapid spread of the analytical technique Thin-Layer Chromatography (TLC). Egon Stahl made TLC known in Europe and standardized it. CAMAG took up the new method and offered as early as 1962 an extensive range of products for the TLC steps used at that time: plate coating, sample application, chromatogram development, reagent transfer for derivatization by spraying and UV inspection.
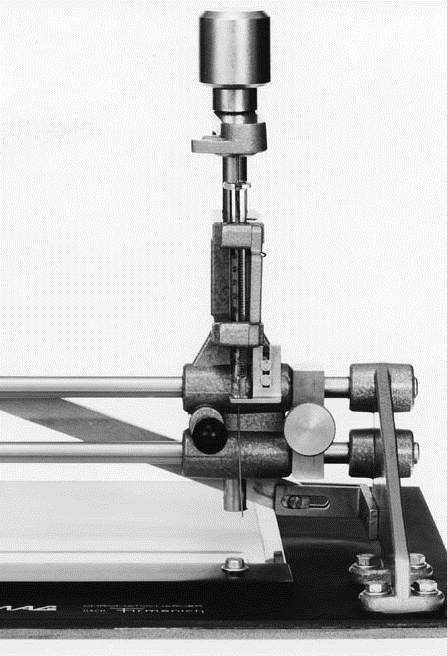
CAMAG® Chromatocharger for application of bands
Remarkable was the CAMAG® Vario-KS chamber according to Geiss and Schlitt (Euratom), which allowed for a systematic optimization of the parameters of the development step. Incidentally, we usually had to "sell" the Vario KS chambers twice, once to the decision-maker to have the money released, and then again to the laboratory staff for use.

CAMAG® Vario-KS chamber according to Geiss and Schlitt
As early as 1964, CAMAG launched its first scanner for the direct photometric evaluation of TLC plates, at the same time as the Zeiss evaluation device developed by Hellmut Jork, which was based on the PMQ-II spectrophotometer. The CAMAG T-Scanner was a Turner fluorometer that was adapted for TLC evaluation by means of a vertically arranged cross measuring stage - today one would call it an interface. By the way, CAMAG soon introduced a corresponding interface for the Zeiss PMQ-II, the Z-Scanner.

CAMAG® T-Scanner (1964)
A very far-sighted decision was to provide TLC customers and interested parties with the relevant TLC literature free of charge. For this purpose, the "CAMAG Bibliography Service" (CBS) was founded in 1965, which made the current TLC publications available in the form of short reviews.
The introduction of the CAMAG® High Voltage Electrophoresis System (HVE) is also part of the first decade of CAMAG's history. Its core was the electrophoresis cell, which was based on a simple but surprisingly effective principle of heat transfer. For almost 30 years the HVE system was an important pillar of the company. We only took it out of the program in the early 80s. On one hand, HPLC had taken over most of the tasks previously solved with HVE, and on the other hand, the now enormously tightened safety regulations would have required a completely new design. However, it should be noted that not a single accident has occurred with a HVE system.
Management Team
Facts and Figures
Founded
1958
Headquarters
Muttenz, Switzerland
Subsidiaries
CAMAG Germany, CAMAG Scientific USA
Distribution
More than 70 distributors serving more than 120 countries; rest of world directly through CAMAG
Customers
Industries, organizations and universities
Quality Management
CAMAG is ISO 9001 certified since 1995 and ISO 9001:2015 since January 2018.
Registration No. 11688.




